Evaluating different web applications to assess the toxicity of
Por um escritor misterioso
Descrição
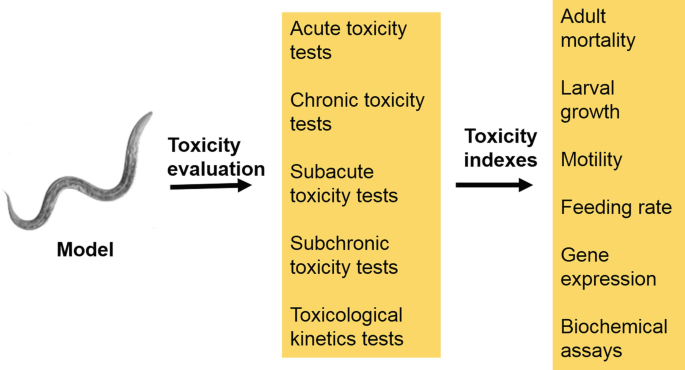
Toxicity Evaluation Using Animal and Cell Models
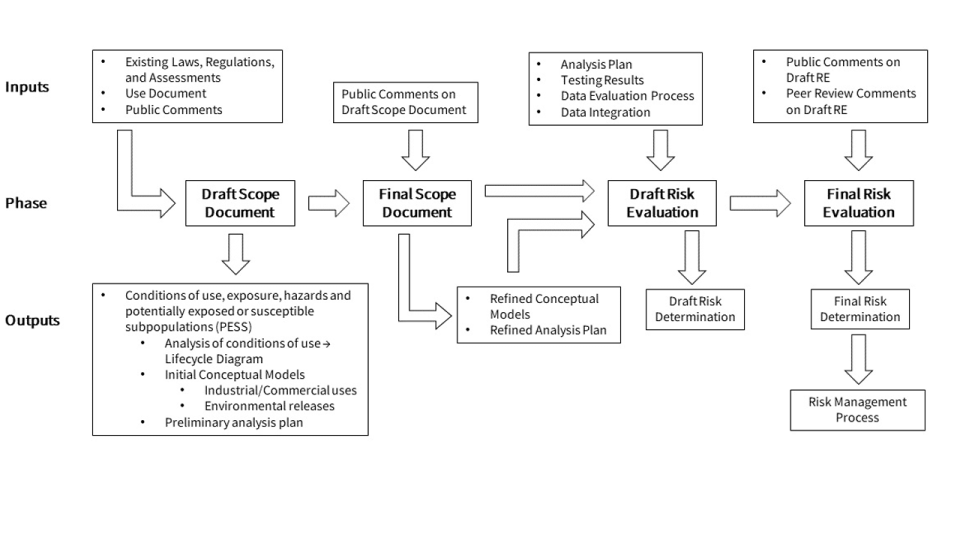
Risk Evaluations for Existing Chemicals under TSCA

The USEtox Model

Systematic evidence map (SEM) template: Report format and methods used for the US EPA Integrated Risk Information System (IRIS) program, Provisional Peer Reviewed Toxicity Value (PPRTV) program, and other “fit for purpose”
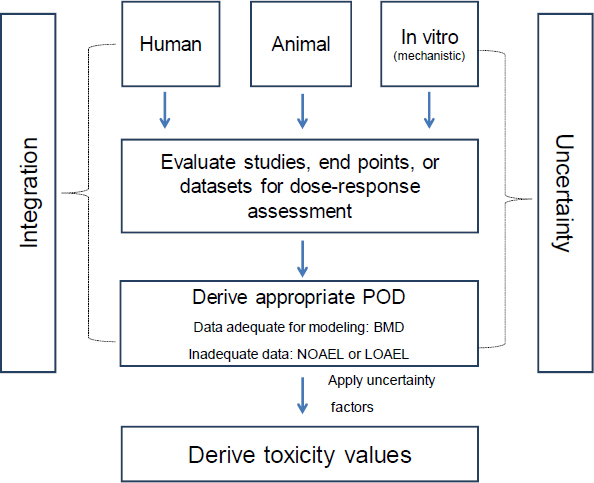
7 Derivation of Toxicity Values, Review of EPA's Integrated Risk Information System (IRIS) Process
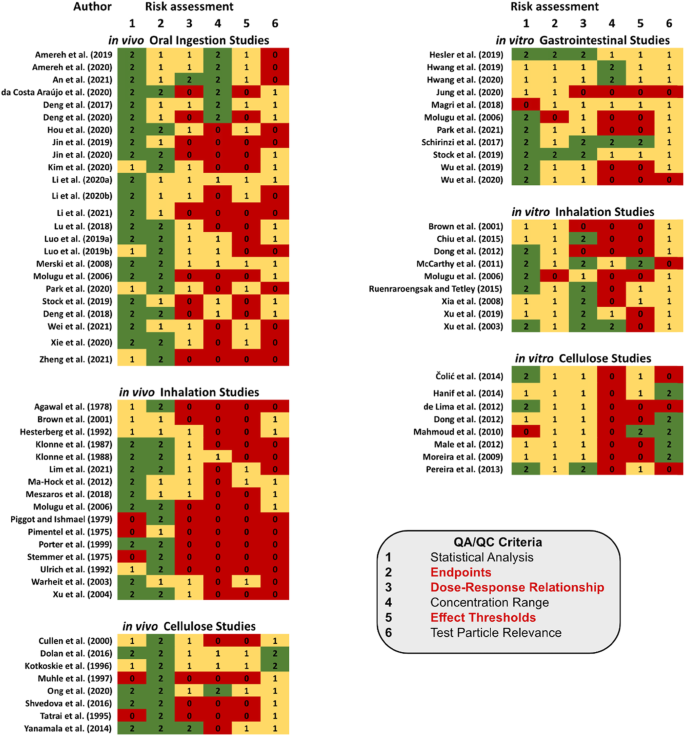
Screening and prioritization of nano- and microplastic particle toxicity studies for evaluating human health risks – development and application of a toxicity study assessment tool, Microplastics and Nanoplastics

Paving the way for application of next generation risk assessment to safety decision-making for cosmetic ingredients - ScienceDirect
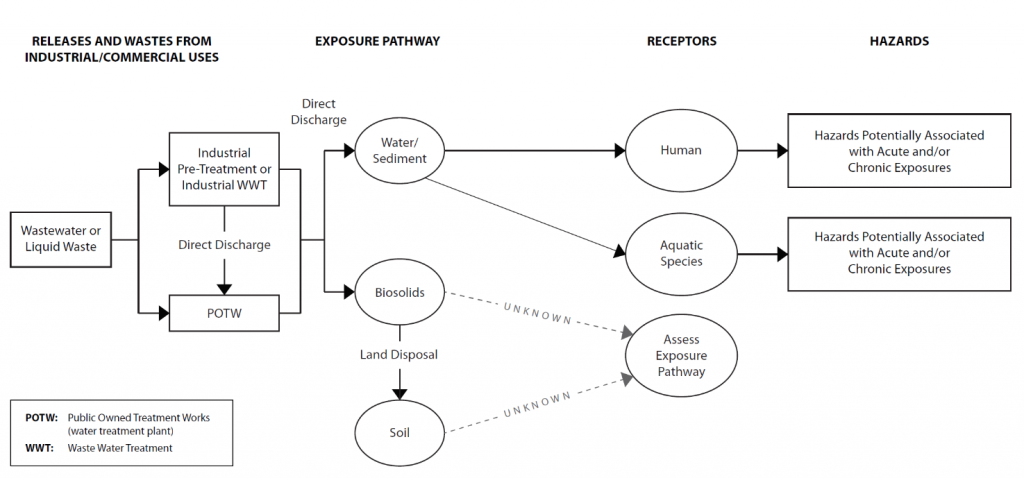
Toxicity and Risk Assessment – 14d
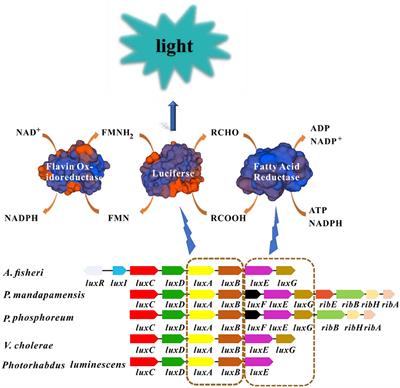
Frontiers Mathematical Modeling Approaches for Assessing the Joint Toxicity of Chemical Mixtures Based on Luminescent Bacteria: A Systematic Review
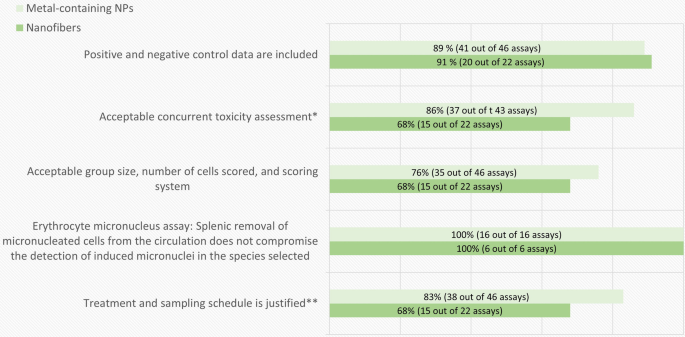
A systematic quality evaluation and review of nanomaterial genotoxicity studies: a regulatory perspective, Particle and Fibre Toxicology
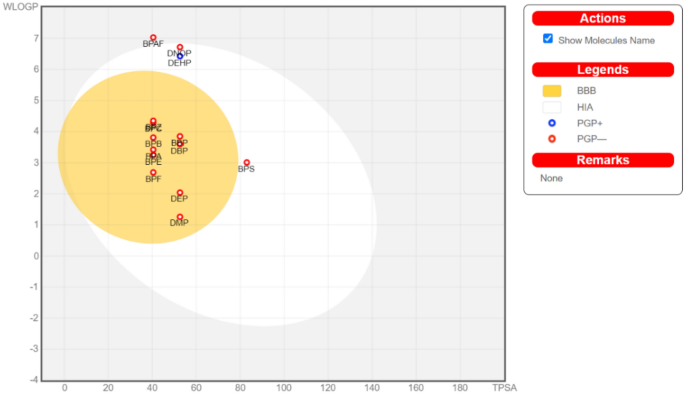
Evaluating different web applications to assess the toxicity of plasticizers

Threshold of Toxicological Concern – ILSI Europe
de
por adulto (o preço varia de acordo com o tamanho do grupo)







